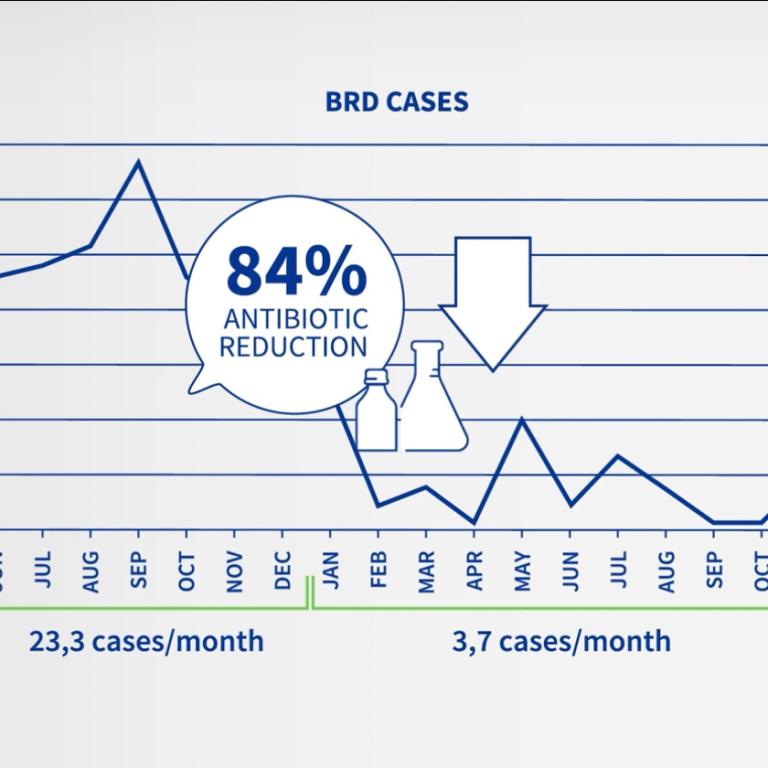
The aim of this study was to confirm the efficacy of a commercial vaccine containing Histophilus somni and Mannheimia haemolytica leukotoxoid (HIPRABOVIS® SOMNI/Lkt) in reducing clinical respiratory signs and the number of concomitant treatments under field conditions.
- Home
- Animal Health
- Knowledge
- Efficacy of a commercial vaccine containing Histop ...
Efficacy of a commercial vaccine containing Histophilus somni and Mannheimia haemolytica leukotoxoid (HIPRABOVIS® SOMNI/Lkt) under field conditions
Related products
Live marker vaccine against BoHV-1 (gE- tk- double-gene deleted) in emulsion for injection
Multivalent viral vaccine against BRSV, BVDV types 1 & 2 and PI3V in emulsion for injection.
Multivalent viral vaccine against BRSV, IBR, BVDV types 1 & 2 and PI3V in emulsion for injection.
Live vaccine against Bovine Respiratory Syncytial Virus (BRSV), in suspension for injection or nasal spray for cattle.
Subunit vaccine against mastitis caused by Streptococcus uberis, in injectable emulsion
Polyvalent inactivated vaccine against bovine mastitis, in injectable emulsion
Inactivated vaccine against pneumonia caused by Histophilus somni and Mannheimia haemolytica leukotoxoid, in injectable emulsion.
Live vaccine (double gene deleted gE-/tk-) against IBR virus, in injectable freeze-dried tablet
Combined vaccine against IBR, PI3, BVD and BRS viruses, in injectable suspension
Inactivated vaccine against infectious bovine keratoconjunctivitis, in injectable suspension
Combined vaccine against PI3, BVD and BRS viruses, in injectable suspension
Inactivated vaccine against enterotoxaemia, tetanus and blackleg, in injectable suspension
Related services
An innovative service based on data analysis that facilitates decision-making in Cattle
HIPRA Stats is a data analysis service for animal production companies.
This online calculator will allow you to know your farm costs, find out the costs associated with clinical and subclinical mastitis and analyse the production losses associated with mastitis
HIPRA University offers high quality training programmes in strategic areas of interest for professionals
HIPRA has specially developed several online tools to support veterinarians and other Animal Health professionals in their daily work.
Access an advanced diagnostic service with maximum comfort and reliability