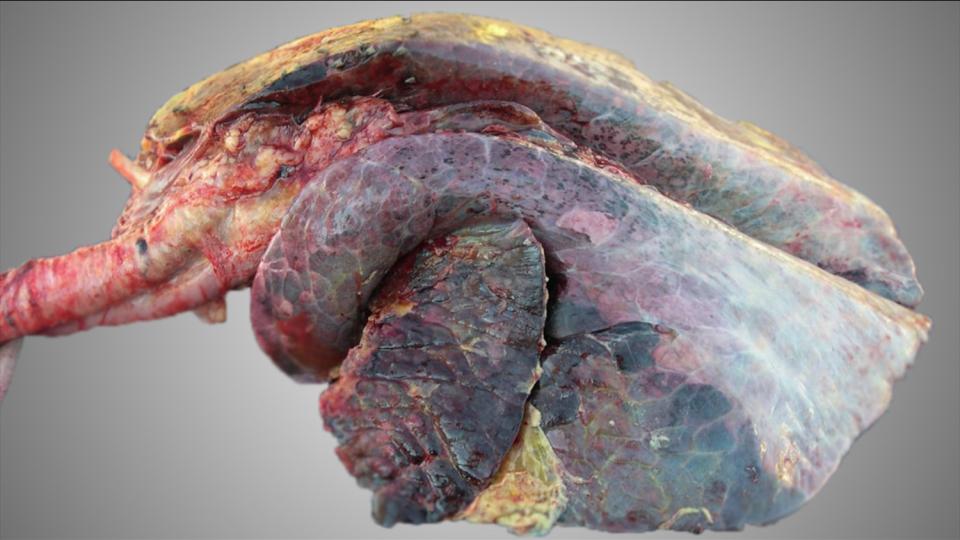
Taking into account that BVDV control, unlike other infectious diseases involved in BRD, is highly feasible in cattle production systems and there are effective tools to break the disease transmission cycle, it is strongly recommended to establish the BVD status of the herd: whether there is evidence of BVD infection.
In dairy herds, screening procedures to establish evidence of active BVD infection are based on the use of serology on a representative number of young animals (9 to 12 or 18 months of age); this approach gives an indication of ongoing or recent infection. If the BVD antibody level is negative in all the samples, it is very likely that they have not been exposed to BVDV recently. PCR can also be done on bulk tank milk for detection of viral circulation within the milking herd. If bulk milk tests positively, BVDV is circulating among the milking cows, but a negative test result is not proof of a BVDV free status.
A second step is identification of individual PI animals on BVDV-positive farms. Testing of pooled blood samples by PCR is a rational means of excluding groups of animals from being BVDV carriers. The latter approach can be performed in combination with PCR on bulk milk as screening procedure in BVDV vaccinated herds. It is advisable to perform sampling and testing for PI animals also in newborns to early identify sources of BVDV infection. Due to colostral antibodies interference with detection of BVDV that might yield false-negative results in calves younger than 4-6 months of age, individual blood samples by PCR or skin biopsies (ear-notch samples) by ELISA may provide reliable test results (Sandvik et al., 2006). BVDV positive animals have to be retested after at least 3 weeks later, in order to rule out transient infection (Schweizer et al., 2021).
The identification and removal of PI animals is considered key to control BVD and have a strongly beneficial impact on the welfare of cattle by preventing transient infections. Central elements of systematic approaches to control and eradication of BVDV have been identified (Lindberg et al., 2006; EFSA AHAW Panel, 2017):
- Biosecurity involves all measures aimed at reducing between-herd transmission, with strong emphasis on preventing contacts with/movements of PI animals, i.e livestock trade, exhibitions and animal contacts on pasture. The efficacy of movement controls is most effective in the context of systematic control at regional and national level. The role of vaccines in systematic control is an additional biosecurity measure; vaccination aims to protect susceptible animals within herd after removal of PI animals or to prevent re-introduction of the infection in free herds, especially in areas where the risk of introducing BVDV infection is known or perceived to be high.
- The second measure is virus elimination by removal of PI animals from infected herds.
- Surveillance is fundamental to monitor the progress of interventions and to rapidly detect new infections.
Author: Camilla Luzzago, DVM, PhD. Associate Professor of Animal Infectious Diseases. Department of Veterinary Medicine and Animal Science. Università degli Studi di Milano, Milan, Italy.
References:
1. Bauermann, F.V., Flores, E.F., Ridpath, J.F. (2012) Antigenic relationships between Bovine viral diarrhea virus 1 and 2 and HoBi virus: possible impacts on diagnosis and control. J Vet Diagn Invest 24:253–261. doi.org/10.1177/1040638711435144
2. Callan, R. J., & Garry, F. B. (2002). Biosecurity and bovine respiratory disease. The Veterinary Clinics of North America. Food Animal Practice, 18(1), 57–77. https://doi.org/10.1016/S0749-0720(02)00004-X
3. Caswell, J.L., Hewson, J. Slavić, D., DeLay, J., Bateman, K., (2012) Laboratory and Postmortem Diagnosis of Bovine Respiratory Disease Vet Clin North Am Food Anim Pract. 28(3): 419–441. doi: 10.1016/j.cvfa.2012.07.004
4. EFSA AHAW Panel (EFSA Panel on Animal Health and Welfare), More S, Bøtner A, Butterworth A, Calistri P, Depner K, Edwards S, Garin-Bastuji B, Good M, Gort_azar Schmidt C, Michel V, Miranda MA, Nielsen SS, Raj M, Sihvonen L, Spoolder H, Stegeman JA, Thulke H-H, Velarde A, Willeberg P, Winckler C, Baldinelli F, Broglia A, Dhollander S, Beltr_an-Beck B, Kohnle L and Bicout D (2017) Scientific Opinion on the assessment of listing and categorisation of animal diseases within the framework of the Animal Health Law (Regulation (EU) No 2016/429): bovine viral diarrhoea (BVD). EFSA Journal 2017;15(8):4952, 45 pp. https://doi.org/10.2903/j.efsa.2017.4952
5. Lindberg A, Brownlie J, Gunn GJ, Houe H, Moennig V, Saatkamp HW, Sandvik T and Valle PS, (2006). The control of bovine viral diarrhoea virus in Europe: today and in the future. Revue Scientifique et Technique (International Office of Epizootics), 25, 961–979.
6. Loneragan, G. H., Thomson, D. U., Montgomery, D. L., Mason, G. L., & Larson, R. L. (2005). Prevalence, outcome, and health consequences associated with persistent infection with bovine viral diarrhea virus in feedlot cattle. Journal of the American Veterinary Medical Association, 226(4), 595–601. https://doi.org/10.2460/javma.2005.226.595
Moennig, V.; Yarnall, M.J. (2021) The Long Journey to BVD Eradication. Pathogens, 10, 1292. https://doi.org/10.3390/pathogens10101292
Pardon, B. & Buczinski, S. (2020) Bovine Respiratory Disease Diagnosis What Progress Has Been Made in Infectious Diagnosis? Vet Clin Food Anim 36, 425–444 https://doi.org/10.1016/j.cvfa.2020.03.005
Sandvik, T., Greiser-Wilke, I., Graham, D. Genome, diagnosis & diagnostic tools (2006) EU Thematic network on BVDV control position paper
Schweizer, M., Stalder, H., Haslebacher, A., Grisiger, M., Schwermer, H., & Di Labio, E. (2021). Eradication of Bovine Viral Diarrhoea (BVD) in Cattle in Switzerland: Lessons Taught by the Complex Biology of the Virus. Frontiers in Veterinary Science, 8, 1012. https://doi.org/10.3389/fvets.2021.702730
Smith, R. A., Step, D.L., Woolums, A. (2020) Bovine Respiratory Disease Looking Back and Looking Forward, What Do We See? Vet Clin Food Anim 36, 239–251 https://doi.org/10.1016/j.cvfa.2020.03.009
Taylor, J. D., Fulton, R. W., Lehenbauer, T. W., Step, D. L., & Confer, A. W. (2010). The epidemiology of bovine respiratory disease: What is the evidence for predisposing factors? The Canadian Veterinary Journal, 51(10), 1095–1102.
Yeşilbağ, K., Alpay, G., Becher, P. (2017) Variability and Global Distribution of Subgenotypes of Bovine Viral Diarrhea Virus. Viruses 9:128. doi.org/10.3390/v9060128